What Are Enzymes? How They Work, Their Types, and Why They Matter
A clear guide to the proteins that power every chemical reaction in your body, from digestion to DNA repair.
What Are Enzymes?
An enzyme is a biological catalyst. It accelerates a specific chemical reaction inside a living cell without being destroyed or permanently altered during that reaction.
Once the reaction finishes, the enzyme is free to repeat the process with a new set of molecules.
Nearly every chemical reaction inside your cells depends on an enzyme to proceed at a speed compatible with life [1].
Your body uses thousands of different enzymes, and scientists have cataloged over 5,000 distinct biochemical reactions that enzymes catalyze [2].
Without these proteins, the reactions that keep you alive, from converting food into energy to copying your DNA, would be so slow they would effectively never happen.
Most enzymes are proteins. A small number of catalytic RNA molecules called ribozymes can also speed up reactions, and researchers identified these in the 1980s [3].
But for nearly all practical purposes, when you hear “enzyme,” you can think “specialized protein with a very specific job.”
“So enzymes are like an on switch for chemical reactions?” – Not exactly.
The reactions they catalyze would eventually happen on their own, given enough time. Enzymes accelerate those reactions, sometimes by factors of millions or more, by lowering the energy barrier required to get the reaction started. That energy barrier is called activation energy, and reducing it is the central trick of every enzyme.
With a definition in place, the next question is what enzymes are physically made of and how their structure determines what they do.
What Are Enzymes Made Of?
Enzymes are proteins built from chains of amino acids, called polypeptides. The human body uses 20 different amino acids, and the specific sequence of those amino acids in a given enzyme determines how the chain folds into a three-dimensional shape [1].
That 3D shape is everything. It dictates which molecules the enzyme can interact with and what reaction it will catalyze.
The most important part of an enzyme’s structure is its active site, a small pocket or groove on the surface of the protein where the target molecule, called the substrate, binds. Only 2 to 4 amino acids in the active site are directly involved in catalysis [4].
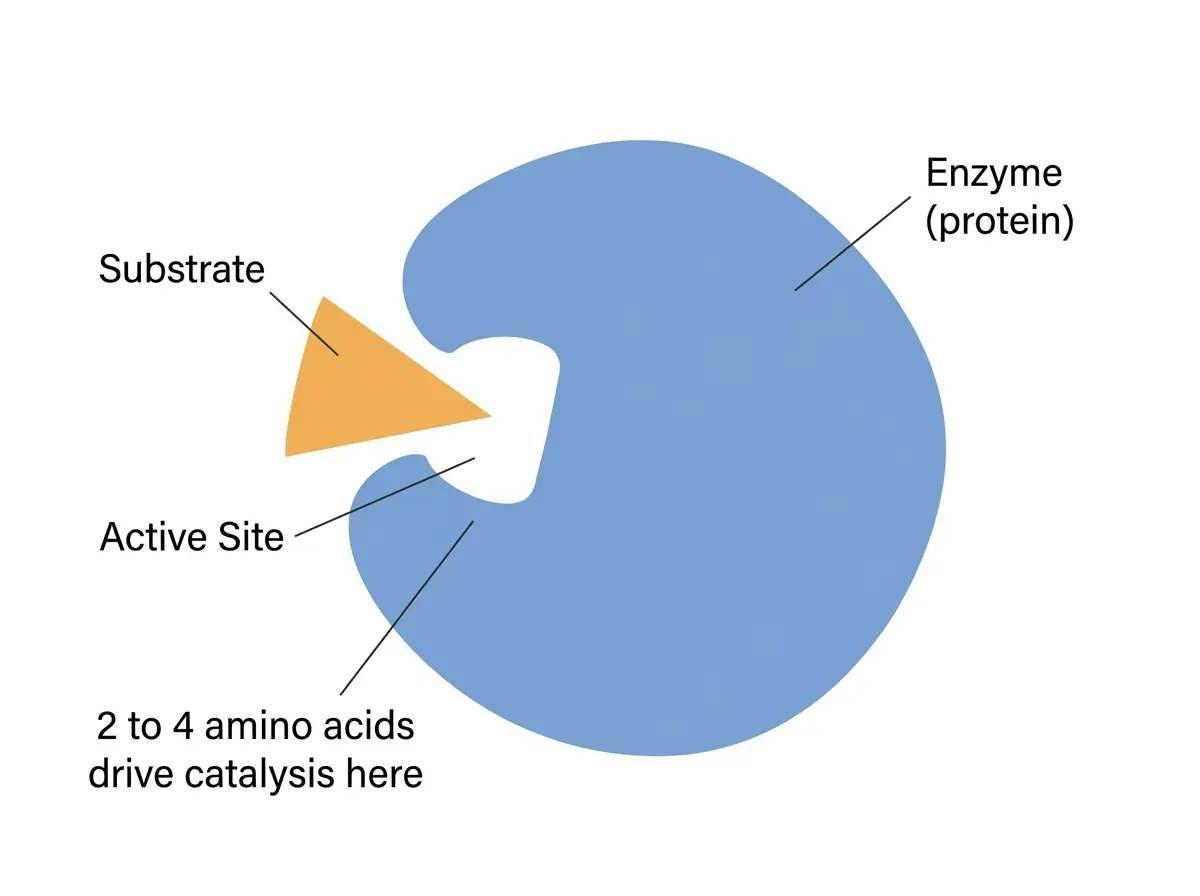
The rest of the enzyme’s structure exists to hold those few amino acids in exactly the right position.
Enzymes vary widely in size. The smallest known enzyme, 4-oxalocrotonate tautomerase, contains only 62 amino acid residues per subunit [5].
The animal fatty acid synthase, on the other end, contains over 2,500 residues [6]. Size does not determine power. What matters is the precise geometry of the active site.
Cofactors and Coenzymes
Some enzymes work perfectly well on their own. Others need a helper molecule to function. These helpers fall into two categories.
Cofactors are inorganic ions, often metals like zinc, iron, magnesium, or copper, that sit within the active site and participate directly in catalysis. Carbonic anhydrase, for example, requires a zinc ion to do its job [7].
Coenzymes are small organic molecules that assist the enzyme, often by carrying chemical groups or electrons between reactions.
Many coenzymes come from vitamins. NAD+ (from niacin/vitamin B3) and FAD (from riboflavin/vitamin B2) are two of the most important coenzymes in your body, both playing central roles in energy metabolism [8].
This connection is one reason dietary vitamins matter so much. Your body cannot synthesize many coenzymes on its own, so it must get the raw materials from food.
An enzyme without its required cofactor or coenzyme is called an apoenzyme. It is inactive. The complete, functional unit, enzyme plus helper molecule, is called a holoenzyme [1].
Structure determines function. The next section explains the specific mechanism by which enzymes accelerate reactions.
How Do Enzymes Work?
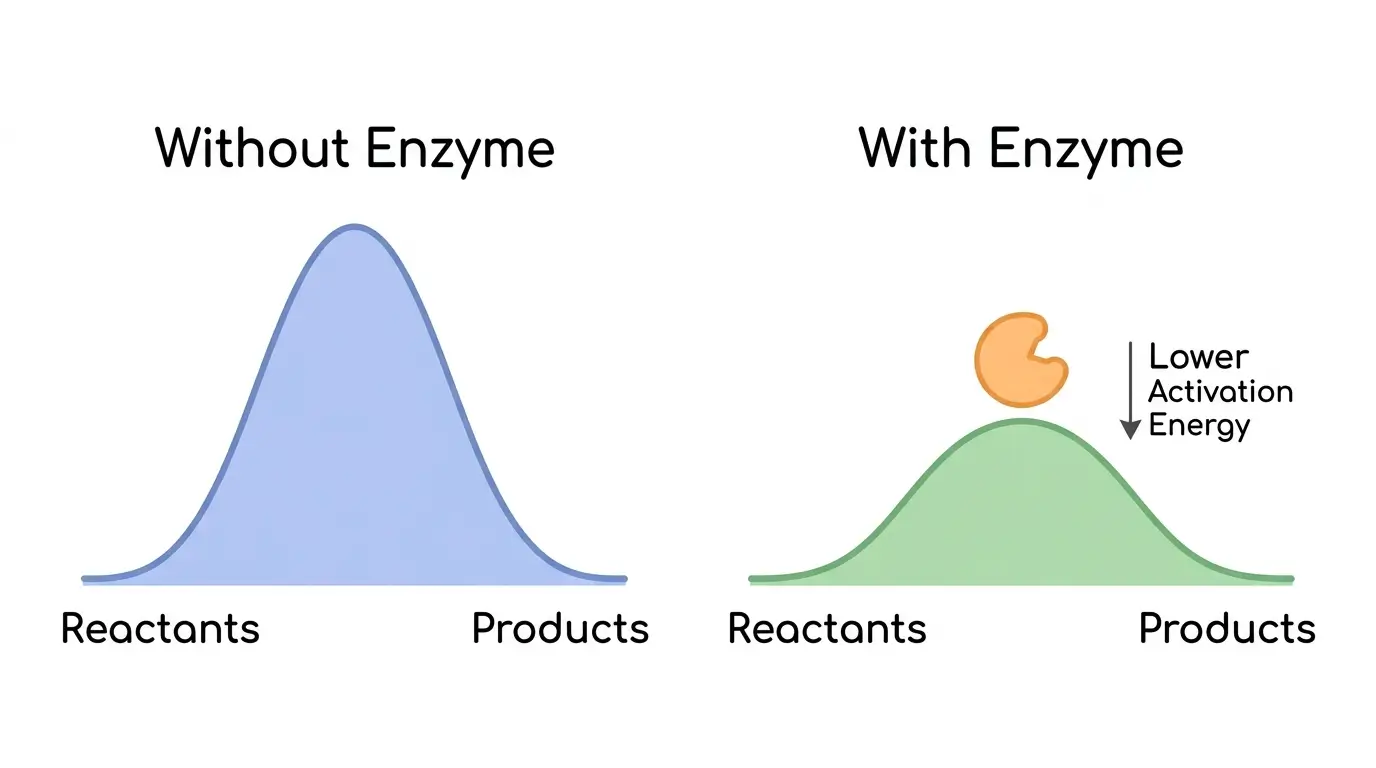
Every chemical reaction requires a minimum amount of energy to get started. That minimum is the activation energy. Think of it as a hill that reactant molecules must climb before they can slide down the other side and become products.
Enzymes lower that hill. They do not change the starting energy of the reactants or the final energy of the products. They do not change the overall equilibrium of the reaction. They simply reduce the energy barrier, making it far easier for the reaction to proceed [1].
How dramatic is this effect? The enzyme orotidine 5’-phosphate decarboxylase accelerates a reaction that would otherwise take an estimated 78 million years to happen on its own. With the enzyme, it occurs in milliseconds [9].
That is a speed increase of roughly 10^17.
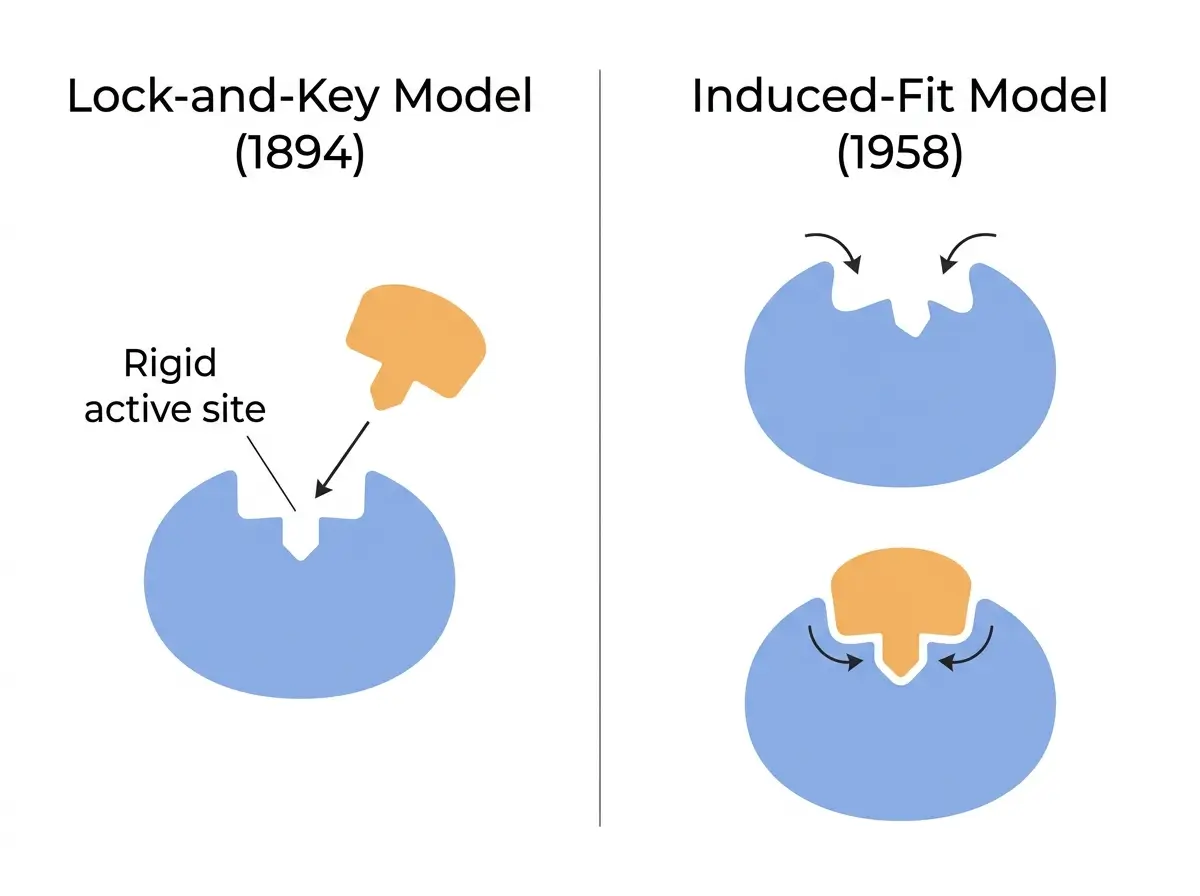
The Lock-and-Key Model
In 1894, German chemist Emil Fischer proposed the first model for how enzymes recognize their substrates. He suggested that the enzyme’s active site has a rigid, fixed shape that perfectly matches the shape of the substrate, similar to how a specific key fits a specific lock [10].
This model explained enzyme specificity well. Each enzyme works on only one substrate (or a narrow group of substrates) because only the correct shape will fit.
The limitation of Fischer’s model is that it treats enzymes as stiff, unchanging structures. Decades of later research showed that proteins are flexible, not rigid.
The Induced-Fit Model
In 1958, Daniel Koshland proposed an updated model. He argued that the active site is not a fixed shape waiting for the perfect substrate. Instead, the enzyme adjusts its shape slightly as the substrate approaches and binds [11].
The active site molds around the substrate, creating a tighter, more effective fit. This is the induced-fit model, and it is the more widely accepted explanation today.
Induced fit also explains why some enzymes can act on a small group of structurally similar substrates. The flexibility allows a degree of accommodation, though the enzyme remains highly selective.
What Happens During the Reaction
The process follows a clear sequence. The substrate binds to the enzyme’s active site, forming an enzyme-substrate complex.
Within this complex, the enzyme stabilizes a high-energy intermediate called the transition state, making it easier for chemical bonds to break or form.
The reaction produces one or more product molecules, which then detach from the active site. The enzyme returns to its original shape, ready for the next substrate molecule.
This cycle can repeat with remarkable speed. Carbonic anhydrase, the enzyme in your red blood cells that converts carbon dioxide into bicarbonate, processes about 600,000 substrate molecules per second [12].
Catalase, which breaks down hydrogen peroxide (a toxic byproduct of metabolism), handles about 93,000 molecules per second [12].
Scientists call this measure the turnover number.
Enzymes are grouped into categories based on the type of reaction they catalyze. Understanding these categories clarifies the range of work enzymes perform.
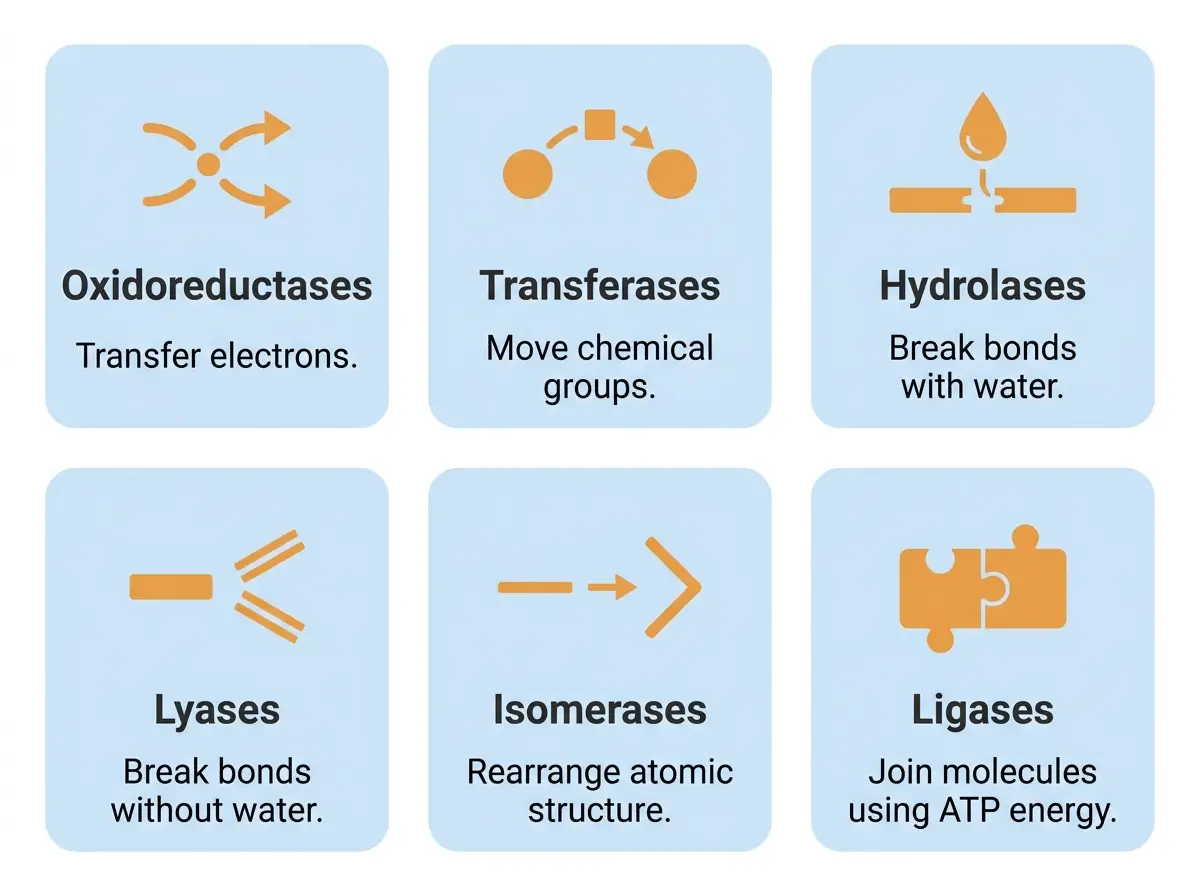
The 6 Major Types of Enzymes (EC Classification)
The International Union of Biochemistry and Molecular Biology classifies all known enzymes using the Enzyme Commission (EC) numbering system [13].
Every enzyme receives a four-digit EC number. The first digit identifies which of six major classes the enzyme belongs to, based on the reaction it catalyzes. The remaining digits narrow the classification further.
Over 5,000 enzymes have been assigned EC numbers to date [2].
| EC Class | Reaction Type | Common Example | Where You Find It |
|---|---|---|---|
| EC 1, Oxidoreductases | Electron transfer | Alcohol dehydrogenase | Liver |
| EC 2, Transferases | Group transfer | Hexokinase | Most cells (glycolysis) |
| EC 3, Hydrolases | Bond breaking with water | Lipase, amylase, protease | Digestive tract, pancreas |
| EC 4, Lyases | Bond breaking (no water) | Aldolase | Most cells (glycolysis) |
| EC 5, Isomerases | Structural rearrangement | Glucose isomerase | Food manufacturing |
| EC 6, Ligases | Joining molecules (uses ATP) | DNA ligase | Cell nucleus |
Many of these enzyme types operate directly inside your body. The next section covers how they keep you alive.
What Do Enzymes Do in Your Body?
Digestive Enzymes
Your digestive system relies on a series of enzymes, each specialized for a different nutrient and active in a different part of your gastrointestinal tract.
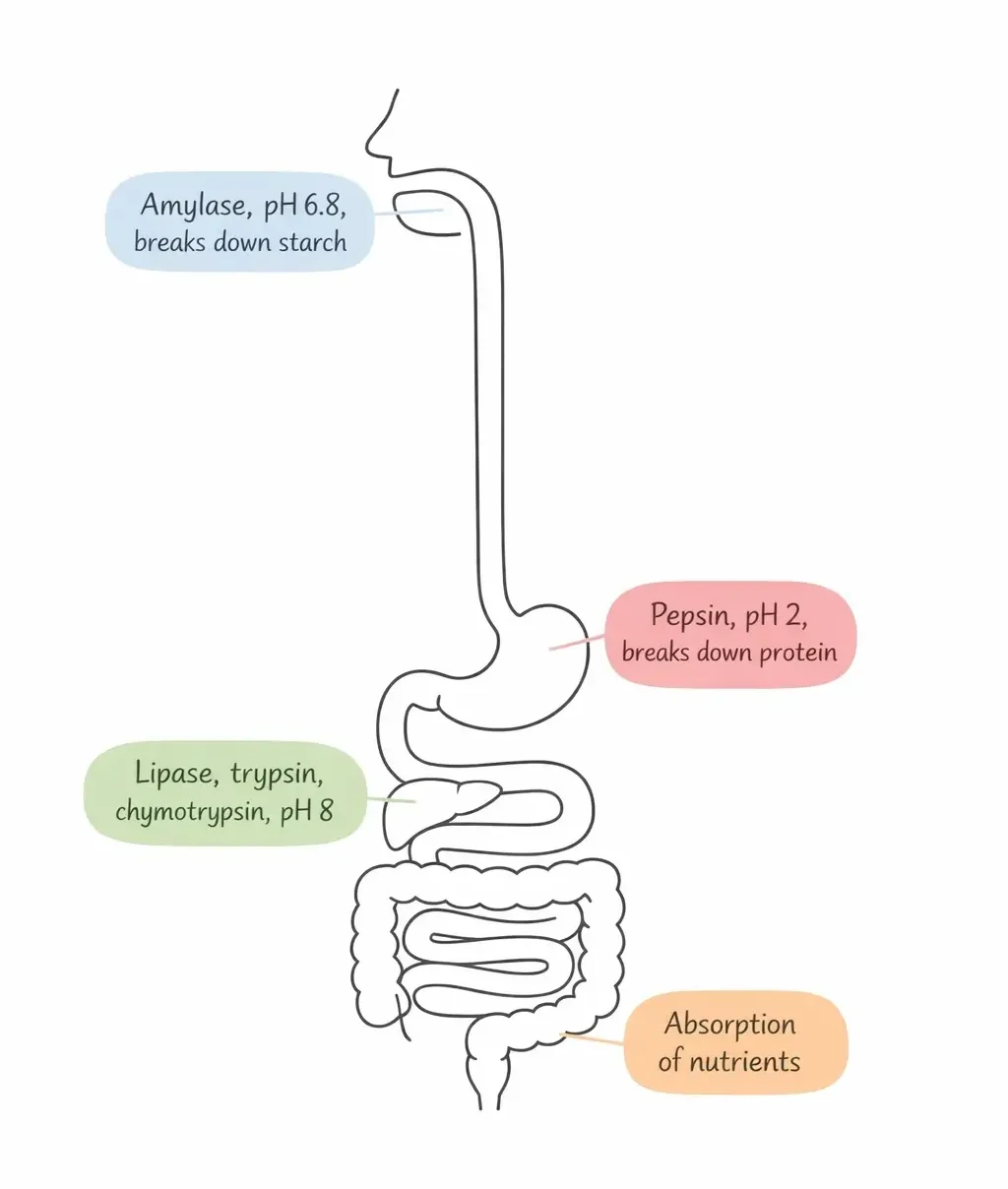
- Amylase, produced in your salivary glands and pancreas, starts breaking down starch into simpler sugars the moment food enters your mouth.
- Pepsin, released in your stomach, tackles proteins, but it works only in the highly acidic environment of gastric juice (around pH 2).
- Your pancreas produces lipase, which breaks down dietary fats in your small intestine, along with trypsin and chymotrypsin, which continue the work of protein digestion at a more alkaline pH (around pH 8) [1].
- Lactase, produced in the lining of your small intestine, splits lactose (milk sugar) into glucose and galactose. If your body produces insufficient lactase, undigested lactose ferments in your gut, causing bloating, gas, and diarrhea. This is lactose intolerance, and it affects roughly 68% of the world’s population to some degree [16].
Why do different enzymes work in different locations? Because each enzyme has a specific pH at which it performs best. Pepsin would denature and stop working in the alkaline environment of your small intestine, and trypsin would fail in the acid of your stomach. Your body compartmentalizes enzyme activity by controlling the chemical environment in each organ.
Metabolic Enzymes
Beyond digestion, enzymes drive the chemical machinery inside every cell. ATP synthase produces adenosine triphosphate, the energy currency your cells use for nearly every task. DNA polymerase copies your genetic code every time a cell divides, with an error rate of less than 1 in 100 million base pairs in high-fidelity versions [1].
Cytochrome P450 enzymes in your liver process and detoxify drugs, hormones, and environmental chemicals.
Other Functions
Carbonic anhydrase in your red blood cells converts CO2 (a waste product of metabolism) into bicarbonate for transport to your lungs, where the process reverses so you can exhale it.
Thrombin activates fibrin to form blood clots when you are injured. Lysozyme, found in your tears, saliva, and nasal mucus, breaks down the cell walls of bacteria and serves as a frontline immune defense [17].
Enzyme performance depends on the conditions they operate in. Changes in temperature or pH can shut enzymes down entirely.
What Affects Enzyme Activity?
Temperature
Most human enzymes work best at your normal body temperature, around 37C (98.6F). As temperature rises, molecular motion increases, and enzyme reactions speed up, but only to a point.
Once the temperature exceeds the enzyme’s tolerance, the protein begins to unfold. This process is called denaturation, and it destroys the precise 3D shape of the active site. A denatured enzyme cannot bind its substrate and stops working [1].
This is one reason a high fever is dangerous. At temperatures above 40C (104F), some of your enzymes begin to lose function, which can disrupt critical metabolic processes.
Not all organisms share this limitation. Bacteria living in volcanic hot springs produce enzymes that remain stable at 80C to 100C. Taq polymerase, isolated from the thermophilic bacterium Thermus aquaticus, works at around 72C and is the enzyme that makes PCR (polymerase chain reaction) technology possible [18]. PCR is the method used in DNA testing, forensic science, and infectious disease diagnosis worldwide.
pH
Each enzyme has a specific pH at which it performs best. Pepsin thrives at pH 2. Trypsin functions near pH 8. Alkaline phosphatase works best around pH 10 [12].
Move too far from that ideal range in either direction, and the charges on the amino acids in the active site change, weakening substrate binding and reducing catalytic speed. Extreme pH values can cause irreversible denaturation.
Your body exploits this sensitivity. By maintaining a pH of about 2 in your stomach and about 8 in your small intestine, it controls which enzymes are active where.
Enzyme Inhibitors
An enzyme inhibitor is any molecule that reduces or blocks enzyme activity. Inhibitors fall into two broad categories.
Competitive inhibitors resemble the substrate and physically block the active site. Malonate, for example, competes with succinate for the active site of succinate dehydrogenase [12].
If enough substrate is present, it can outcompete the inhibitor and restore normal enzyme function.
Non-competitive inhibitors bind to a different location on the enzyme (an allosteric site), causing a shape change that distorts the active site. Because they do not compete with the substrate for the same binding spot, adding more substrate does not overcome their effect.
Many important drugs work as enzyme inhibitors. Aspirin irreversibly inhibits cyclooxygenase (COX), which reduces inflammation and pain [19].
Statins inhibit HMG-CoA reductase, the enzyme your liver uses to produce cholesterol [20].
ACE inhibitors block angiotensin-converting enzyme, lowering blood pressure.
On the harmful side, cyanide is a potent enzyme inhibitor. It blocks cytochrome c oxidase, the final enzyme in your mitochondrial electron transport chain, which halts ATP production and can cause death within minutes [1].
Substrate Concentration
Increasing the amount of substrate available speeds up an enzyme’s reaction rate, but only up to a point. Once every active site on every available enzyme molecule is occupied, the reaction hits its maximum velocity (known as Vmax). Adding more substrate beyond this point has no additional effect because the enzyme is saturated [12].
When enzymes are missing or defective, the consequences range from mild discomfort to serious disease.
What Happens When Enzymes Don’t Work Properly?
Enzyme deficiencies are often genetic. A mutation in the gene encoding a specific enzyme can reduce the enzyme’s activity or prevent the body from producing it at all. The results depend on which enzyme is affected and how central it is to normal metabolism.

- Lactose intolerance is the most widespread example. Reduced production of lactase affects approximately 68% of the global population, with the highest rates in East Asian, West African, and Mediterranean populations [16]. It causes digestive symptoms but is manageable by avoiding dairy or taking lactase supplements.
- Phenylketonuria (PKU) results from a deficiency of phenylalanine hydroxylase, the enzyme that converts the amino acid phenylalanine into tyrosine. Without it, phenylalanine accumulates to toxic levels and can cause severe intellectual disability if untreated. Newborn screening programs detect PKU in the first days of life, and a strict low-phenylalanine diet prevents damage [21].
- G6PD deficiency is the most common enzyme deficiency worldwide, affecting an estimated 400 million people [22]. It impairs the enzyme glucose-6-phosphate dehydrogenase, which protects red blood cells from oxidative damage. Certain triggers, including specific medications, fava beans, and infections, can cause episodes of hemolytic anemia (rapid destruction of red blood cells).
- Gaucher disease involves a deficiency of glucocerebrosidase, leading to the buildup of fatty substances in the spleen, liver, and bone marrow [23]. Enzyme replacement therapy, delivered intravenously, is the standard treatment.
- Fabry disease results from a deficiency of alpha-galactosidase A. Fatty deposits accumulate in blood vessel walls, kidneys, and the heart, causing pain, kidney failure, and cardiovascular complications [24].
- Exocrine pancreatic insufficiency (EPI) occurs when your pancreas does not produce enough digestive enzymes. Chronic pancreatitis, pancreatic cancer, and cystic fibrosis are common causes. People with EPI cannot properly break down food or absorb nutrients, leading to malnutrition, weight loss, and fatty stools [25].
Doctors also use enzyme levels in the blood as diagnostic tools. Elevated ALT and AST (liver enzymes) can signal liver damage or disease [26].
Elevated troponin (a protein released when heart muscle cells die) is one of the primary markers used to diagnose a heart attack.
For some of these conditions, enzyme replacement or supplementation is the primary treatment.
Do Enzyme Supplements Work?
The answer depends entirely on who is taking them and why.
Prescription enzyme replacement therapy is well-supported by clinical evidence. Pancrelipase (brand names include Creon and Zenpep) is FDA-approved for people with EPI and contains a standardized mix of lipase, protease, and amylase. Clinical trials show that it significantly improves fat absorption and reduces symptoms of maldigestion [25].
Lactase supplements (such as Lactaid) are effective for people with lactose intolerance. Taking a lactase pill before consuming dairy helps break down lactose and prevents symptoms in most users [16].
Over-the-counter (OTC) digestive enzyme supplements are a different story. These products are classified as dietary supplements, not drugs, and the FDA does not evaluate them for efficacy before they reach store shelves [27].
Marketing claims often promise relief from bloating, gas, and general digestive discomfort, but the evidence supporting these benefits in healthy individuals is limited.
Johns Hopkins Medicine states directly that most healthy people do not need digestive enzyme supplements [28].
Harvard Health Publishing reached a similar conclusion, noting that OTC enzyme supplements may help some people with specific food intolerances but are unlikely to benefit the general population [29].
“Should I take a digestive enzyme supplement?”
- If you have a diagnosed enzyme deficiency or a condition like EPI, the answer is likely yes, under medical supervision.
- If you are generally healthy and experience occasional bloating, the evidence does not support routine supplementation. Supporting your gut bacteria through probiotics may be a more evidence-based approach for general digestive comfort
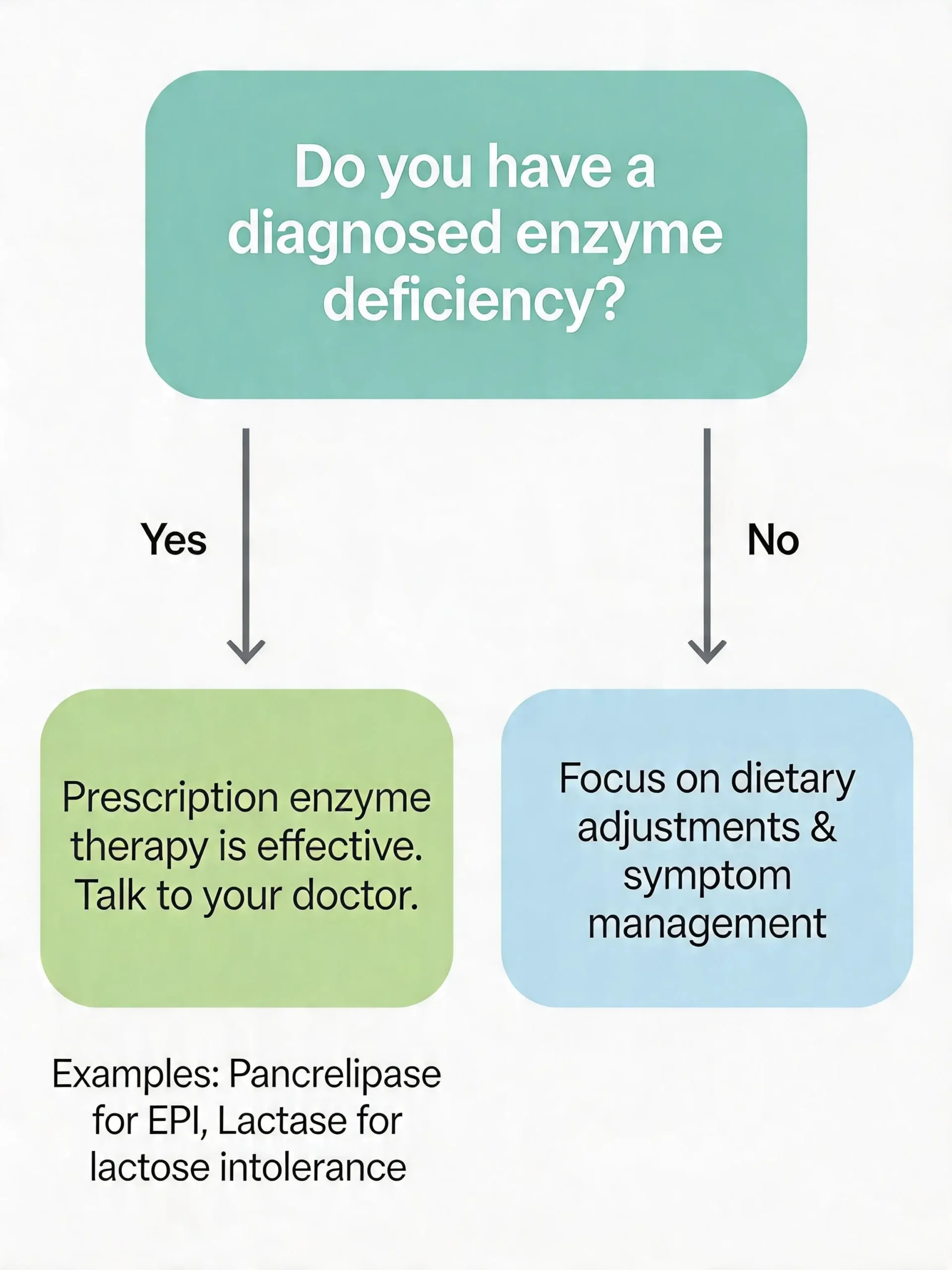
Talk to your doctor before starting any enzyme product.
One of the best Enzyme supplements in the market is MassZymes by BiOptimizers. We reviewed the supplement if you want to take a deeper look or you can visit their sales page directly.
Beyond the human body, enzymes have a wide range of practical uses in industry, medicine, and everyday products.
Enzymes in Industry and Everyday Life
Enzymes are a multibillion-dollar commercial industry. The global industrial enzyme market was valued at approximately $7.1 billion in 2023 and continues to grow [30].
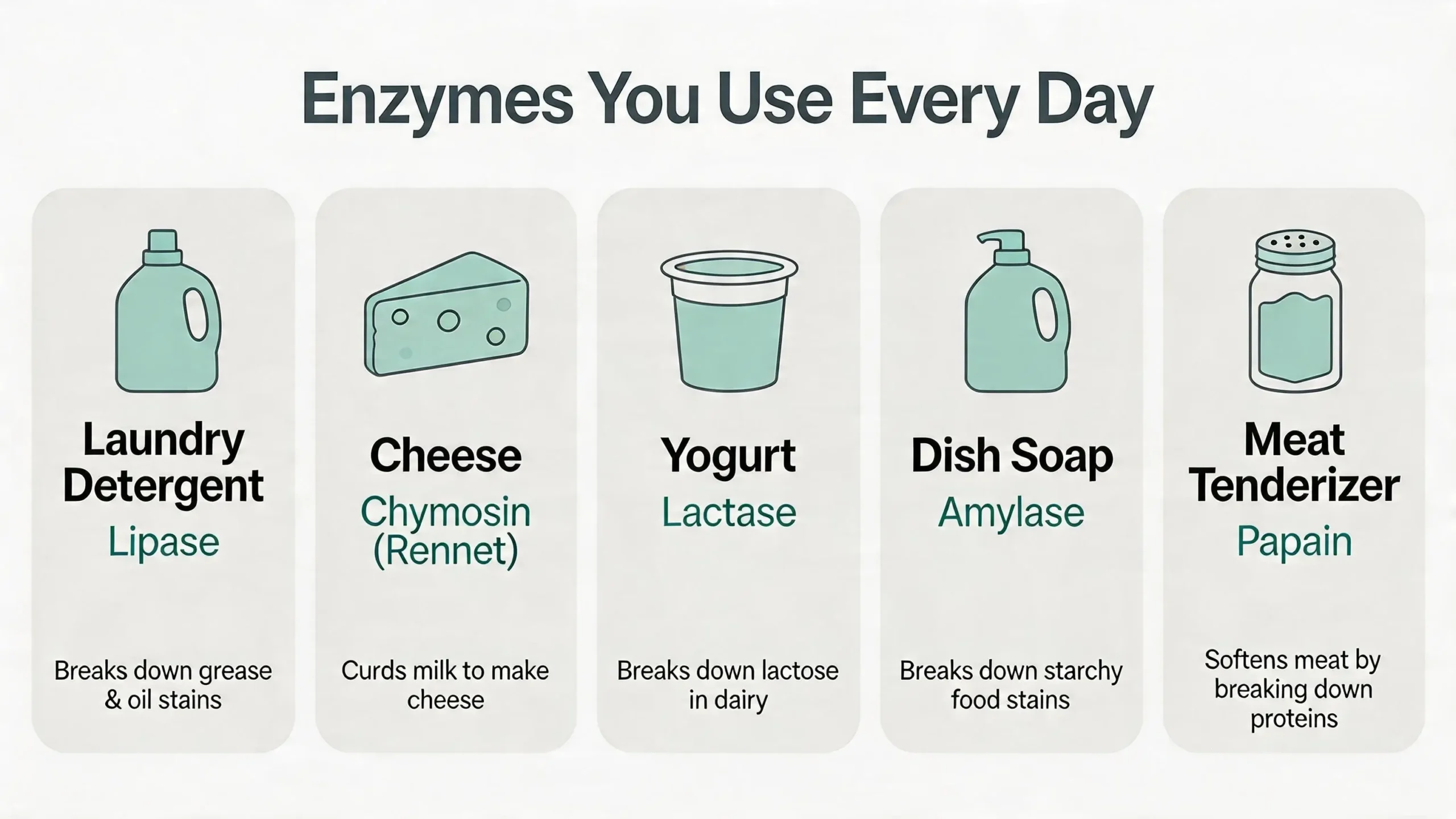
- Laundry detergents were among the first consumer products to use enzymes. Modern detergents contain protease (protease breaks down proteins into amino acids), amylase (to break down starchy food stains), and lipase (to break down grease and oil). These enzymes allow effective cleaning at lower water temperatures, which saves energy [31].
- Food production relies heavily on enzymes. Chymosin (rennet) curdles milk during cheese-making. Amylase converts starch into sugars during bread baking, improving texture and browning. Pectinase clarifies fruit juices by breaking down cell wall components. Glucose isomerase converts glucose into fructose in the production of high-fructose corn syrup [15].
- Medicine uses enzymes in several ways beyond supplements. Taq polymerase powers the PCR process, which underpins COVID-19 testing, paternity testing, forensic identification, and genetic disease screening [18]. Streptokinase and tissue plasminogen activator (tPA) dissolve dangerous blood clots during heart attacks and strokes.
- Biofuel production depends on cellulase enzymes that break down plant cellulose into fermentable sugars, which are then converted into ethanol [32]. This process is central to the development of second-generation biofuels made from agricultural waste rather than food crops.
- Textile manufacturing uses enzymes for bio-polishing cotton fabrics (cellulase smooths the surface) and for removing starch-based sizing agents (amylase) applied during weaving [31].
What connects all these applications is the same principle that operates inside your cells. Enzymes catalyze specific reactions with high efficiency, under mild conditions, and without producing harmful byproducts.
Frequently Asked Questions About Enzymes
How We Researched This
This article was developed by reviewing over 25 sources, including peer-reviewed research papers, clinical reference materials, and educational resources from institutions such as the National Institutes of Health (Genome.gov), the National Center for Biotechnology Information (PMC), Cleveland Clinic, Johns Hopkins Medicine, Harvard Health Publishing, Khan Academy, Britannica, and the American Chemical Society.
Sources were selected based on institutional authority, peer-review status, recency (data covers findings through early 2025), and relevance to the target audience.
Priority was given to peer-reviewed journal articles, established biochemistry textbooks (particularly Stryer’s Biochemistry, which multiple top-ranking sources also cite), and clinical guidance from major academic medical centers. Health-related claims reference published clinical guidance or systematic reviews.
This article focuses on general principles, the most commonly encountered enzyme types, and the health and industrial applications most relevant to non-specialist readers.
It does not cover enzyme kinetics equations (such as the full Michaelis-Menten derivation) in mathematical detail, nor does it address emerging research on artificial enzymes, directed evolution, or computational enzyme design beyond a surface level.
Readers seeking advanced enzymology content should consult the PMC and textbook sources listed in the citations below. All statistics have been attributed to specific, verifiable sources.
Where exact figures vary across sources (e.g., estimated number of enzymes in the human body), we have reported the most commonly cited range.
References
- [1] Stryer, L., Berg, J.M., Tymoczko, J.L. “Biochemistry” – W.H. Freeman, 8th Edition (referenced sections: 8.1, 5.3.1, 10.3, 17)
- [2] Enzyme entry – “Enzyme” (Wikipedia, citing Bairoch, A.) – Updated 2025 – References: “Enzymes are known to catalyze over 5,000 types of biochemical reactions.”
- [3] Cech, T.R. “The Ribosome Is a Ribozyme” – Science, 2000 – DOI: 10.1126/science.289.5481.878
- [4] Suzuki, H. “How Enzymes Work: From Structure to Function” – Referenced in Essays in Biochemistry, 2015 – PMC4692135
- [5] Stivers, J.T. et al. – 4-Oxalocrotonate tautomerase structure – Referenced in Stryer Biochemistry and Wikipedia Enzyme article.
- [6] Maier, T. et al. – Fatty acid synthase structure – Referenced in Stryer Biochemistry and Wikipedia Enzyme article.
- [7] Lindskog, S. “Structure and Mechanism of Carbonic Anhydrase” – Pharmacology and Therapeutics, 1997 – PMID: 9261083
- [8] Lienhart, W.D. et al. “The Human Flavoproteome” – Archives of Biochemistry and Biophysics, 2013 – PMID: 23416055
- [9] Radzicka, A. and Wolfenden, R. “A Proficient Enzyme” – Science, 1995 – PMID: 7489718
- [10] Fischer, E. – “Einfluss der Configuration auf die Wirkung der Enzyme” – Berichte der Deutschen Chemischen Gesellschaft, 1894
- [11] Koshland, D.E. “Application of a Theory of Enzyme Specificity to Protein Synthesis” – PNAS, 1958 – DOI: 10.1073/pnas.44.2.98
- [12] Palmer, T. and Bonner, P.L. – “Enzymes: Biochemistry, Biotechnology, Clinical Chemistry” – Referenced via Essays in Biochemistry (PMC4692135), 2015 – Turnover rates table and kinetic data.
- [13] International Union of Biochemistry and Molecular Biology. “Enzyme Nomenclature” – ExpASy Enzyme Database
- [14] Infinita Biotech. “What Are Enzymes And How Do They Work?” – Referenced for enzyme class distribution data.
- [15] Bhosale, S.H. et al. “Molecular and Industrial Aspects of Glucose Isomerase” – Microbiological Reviews, 1996 – PMID: 8830261
- [16] Storhaug, C.L. et al. “A Systematic Review of the Global Prevalence of Lactose Malabsorption” – Scandinavian Journal of Gastroenterology, 2017 – PMID: 28053336
- [17] Genome.gov – National Human Genome Research Institute. “Enzyme” – Genetics Glossary, 2025
- [18] Saiki, R.K. et al. “Primer-Directed Enzymatic Amplification of DNA with a Thermostable DNA Polymerase” – Science, 1988 – PMID: 2448875
- [19] Vane, J.R. and Botting, R.M. “The Mechanism of Action of Aspirin” – Thrombosis Research, 2003 – PMID: 12804452
- [20] Istvan, E.S. and Deisenhofer, J. “Structural Mechanism for Statin Inhibition of HMG-CoA Reductase” – Science, 2001 – PMID: 11349148
- [21] National Institutes of Health. “Phenylketonuria (PKU)” – MedlinePlus, 2024
- [22] Cappellini, M.D. and Fiorelli, G. “Glucose-6-Phosphate Dehydrogenase Deficiency” – The Lancet, 2008 – PMID: 18061065
- [23] Grabowski, G.A. “Phenotype, Diagnosis, and Treatment of Gaucher’s Disease” – The Lancet, 2008 – PMID: 18971340
- [24] Cleveland Clinic. “Fabry Disease” – Health Articles, 2024
- [25] Cleveland Clinic. “What Are Enzymes, Pancreas, Digestion & Liver Function” – Health Articles, 2024
- [26] Cleveland Clinic. “Elevated Liver Enzymes” – Health Symptoms, 2024
- [27] U.S. Food and Drug Administration. “Dietary Supplements” – FDA.gov
- [28] Johns Hopkins Medicine. “Digestive Enzymes and Digestive Enzyme Supplements” – Health Wellness, 2024
- [29] Harvard Health Publishing. “Gut Reaction: A Limited Role for Digestive Enzyme Supplements” – 2023
- [30] Grand View Research. Industrial Enzymes Market Size, 2023 estimate. (Commonly cited across industry reports; approximate figure.)
- [31] Kirk, O. et al. “Industrial Enzyme Applications” – Current Opinion in Biotechnology, 2002 – PMID: 12069681
- [32] Bhat, M.K. “Cellulases and Related Enzymes in Biotechnology” – Biotechnology Advances, 2000 – PMID: 10704994
- [33] American Chemical Society. “Enzymes: Moving at the Speed of Life” – Celebrating Chemistry, 2021
You can share and adapt this content for non-commercial purposes, provided you give appropriate credit.
