Food Noise Explained: The Neuroscience of Constant Hunger and Why GLP-1s Quiet It
Food noise isn't lack of willpower — it's neural reward activity. Here's what the research says about why GLP-1 drugs consistently silence it.
Until recently, “food noise” wasn’t a clinical term. Patients would describe it to their doctors in roundabout ways – intrusive thoughts about food, constant snacking despite not being hungry, difficulty concentrating because their mind kept returning to what they might eat next.
The experience was real, but the vocabulary didn’t exist.
Then GLP-1 receptor agonists like semaglutide and tirzepatide arrived in mainstream use. Patients who started these medications began describing something striking: the food noise stopped. Almost overnight.
And once people had a name for what they were losing, they could finally describe what it had been like to live with it.
This article reviews what we know about food noise at the neurobiological level, why GLP-1 drugs appear to silence it, and what the emerging research suggests about who experiences it most intensely.
What Is Food Noise, Physiologically
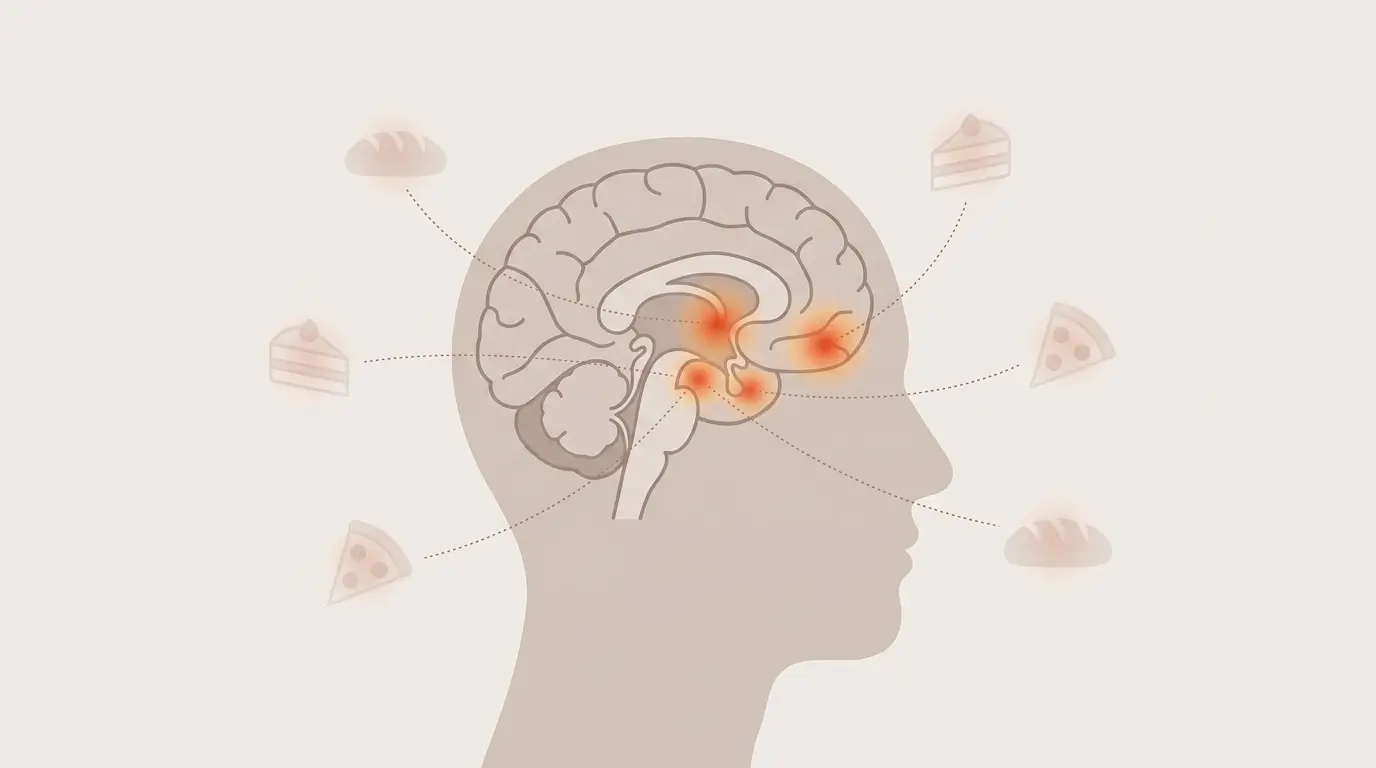
Food noise refers to the persistent, intrusive mental preoccupation with food. It operates largely outside conscious choice.
It shows up as cravings, rumination about past meals, preoccupation with future meals, and difficulty disengaging from food cues in the environment.
Functional MRI studies suggest that food noise correlates with heightened activity in brain regions involved in reward and craving, particularly the ventral striatum, amygdala, and orbitofrontal cortex.
People with higher body mass index tend to show greater activation in these regions when shown food images, even in a non-hungry state.
The practical consequence is that environmental food cues – a bakery smell, a coworker’s lunch, a food commercial – produce stronger and more persistent mental activity in some people than others.
This is not a matter of willpower. It’s baseline neural responsiveness.
The GLP-1 Connection
GLP-1 (glucagon-like peptide-1) is an endogenous hormone released by intestinal cells after eating.
It has multiple actions: slowing gastric emptying, stimulating insulin release, and – critically for this discussion – acting on GLP-1 receptors in the brain, particularly in the hypothalamus and hindbrain.
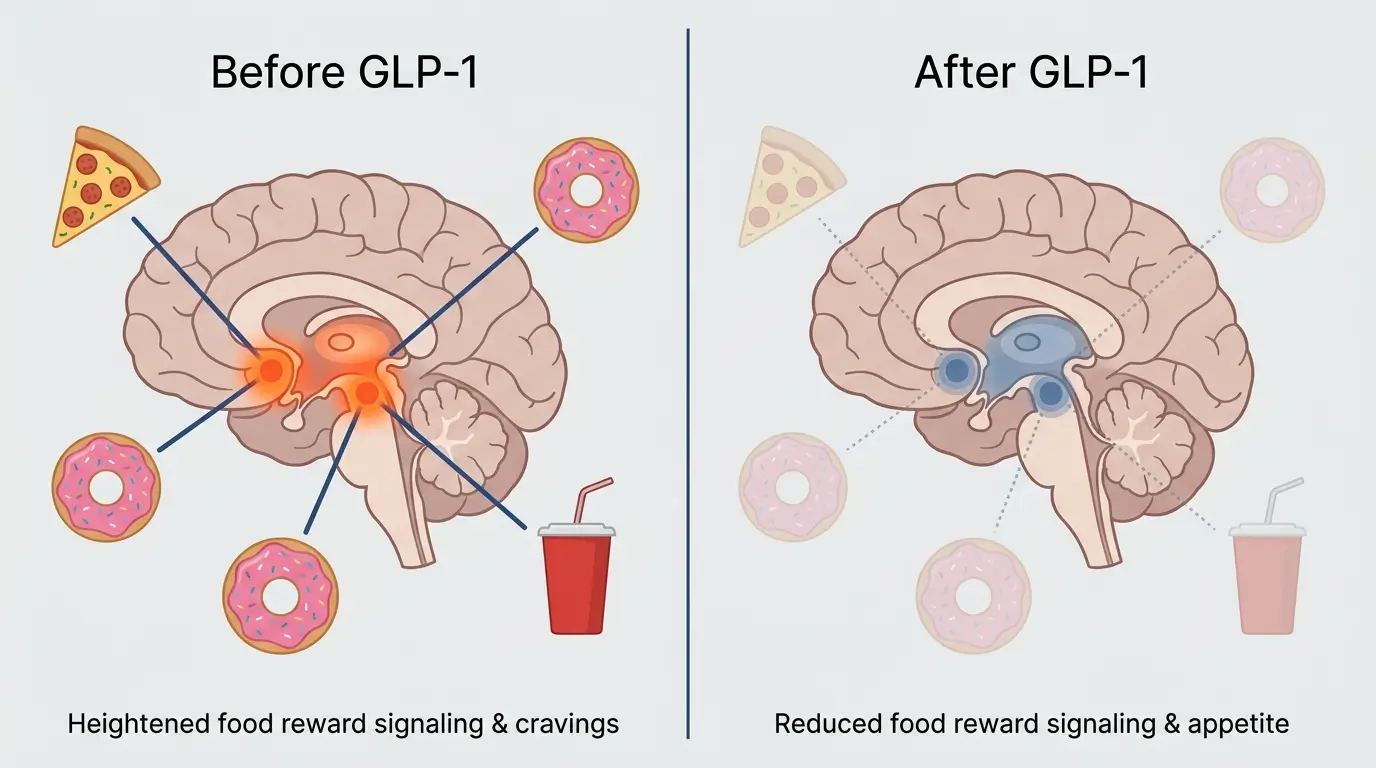
When pharmaceutical GLP-1 receptor agonists are administered, they bind to these same central receptors at much higher levels than endogenous GLP-1 ever achieves.
The result appears to be reduced activity in the reward circuitry that processes food cues. Patients consistently report that food simply becomes less interesting.
Not in a forced way. It fades from mental foreground to mental background, where it seems to belong.
A 2026 study published in Nature analyzing 27,885 GLP-1 users found that genetic variants in GLP1R predicted both the magnitude of weight loss and the severity of nausea side effects.
This suggests that individual variability in food noise response may also have a genetic component, which could partially explain why some people lose weight more easily on these medications than others.
Why This Matters for the Broader Conversation
For decades, clinical and public health approaches to obesity assumed that eating behavior was primarily a product of conscious choice modulated by education and willpower. The GLP-1 era is forcing a reconsideration.
If a single medication can consistently silence the intrusive food thoughts that millions of people experience — without changing their knowledge, values, or motivation – then the mental experience of food was never primarily cognitive. It was neurochemical. The cognitive experience was downstream.
This doesn’t invalidate behavior change strategies. Mindful eating, food journaling, and cognitive-behavioral approaches still work.
But they are more effective when the biological background signal is dampened. F
or patients who want to understand how GLP-1 medications interact with the food noise experience specifically, this comprehensive food noise and GLP-1 guide (https://glp1watchdog.com/guides/food-noise) covers both the mechanism and practical implications.
Who Experiences Food Noise Most Intensely
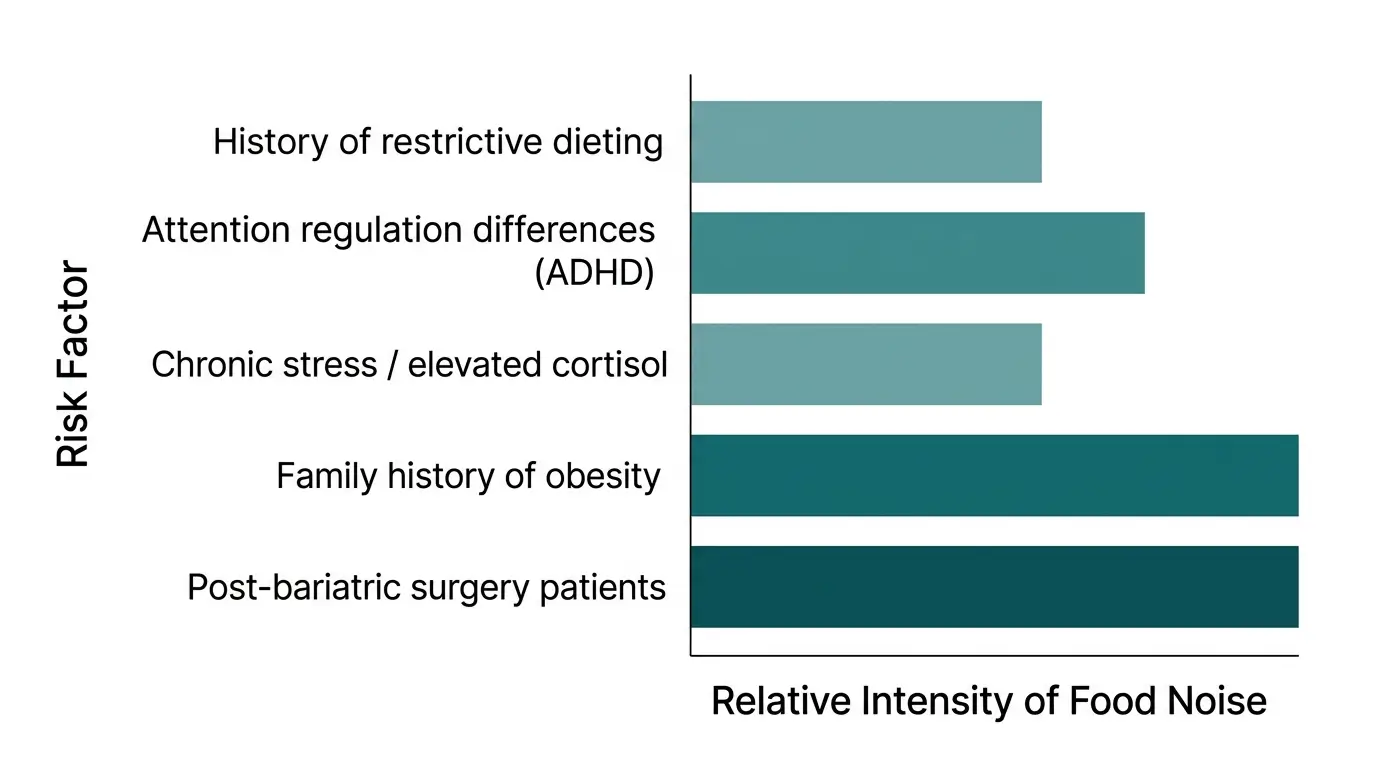
Clinical observation and emerging research suggest several groups are disproportionately affected:
- Individuals with a history of restrictive dieting, which sensitizes reward circuitry
- Those with attention regulation differences, including ADHD, where sustained non-food focus is already taxing
- People under chronic stress, which elevates cortisol and amplifies reward-seeking behavior
- Individuals with a family history of obesity, suggesting a heritable component
- Post-bariatric surgery patients whose hormonal changes did not fully extinguish food preoccupation
These overlapping risk factors explain why weight loss outcomes historically varied so dramatically between individuals following the same diet plan.
What’s Next
The research landscape is moving quickly. Clinical trials are underway for next-generation compounds that target GLP-1, GIP, and glucagon receptors simultaneously.
Oral formulations have made the therapy more accessible. And for the first time, we have a pharmacological tool that acts on the biological substrate of hunger itself rather than trying to outwork it.
Food noise is now a recognized concept in the clinical literature. Patients have the vocabulary to describe what they experience. And medicine has, at last, something to offer them that actually addresses the root — not the symptom.
