KetoCitra Review: Does This Medical Food Actually Help Your Kidneys?
What 30+ user experiences, clinical trial data, and real pricing reveal about this PKD supplement.
Quick Summary
KetoCitra Review: Does This Medical Food Actually Help Your Kidneys?
KetoCitra is a medical food made by Santa Barbara Nutrients that combines beta-hydroxybutyrate (BHB) and citrate to support kidney health in people with chronic kidney disease, primarily polycystic kidney disease (ADPKD). A 2026 interim clinical trial showed a 5% reduction in total kidney volume over 90 days in the treatment group [1]. Most users report stable or improved kidney function when combining it with dietary changes. It costs $139 to $159 per month depending on how you buy it. The biggest limitations are the price, the need for a ketogenic diet for best results, and the early stage of clinical evidence.
Product Brand: KetoCitra
4
Pros
- Backed by UCSB research
- Sodium-free, kidney-safe formula
- 2026 clinical trial shows TKV reduction
- All GRAS-certified ingredients
- Users report GFR stabilization
- Pleasant lemon taste
- No prescription required
Cons
- $139-$159 per month
- Best results require keto diet
- Not for CKD stages 4-5
- Contains potassium (hyperkalemia risk)
- Limited controlled trial data
- Not sold on Amazon
- Founder has financial interest in research
If you’re considering trying it, you can check current pricing and subscription options at SantaBarbaraNutrients.com.
What You Need to Know Before Buying
If you have ADPKD or another form of chronic kidney disease, you’ve probably spent hours searching for anything that might slow your disease progression.
KetoCitra lands in a unique space. It is not a drug. It is not a dietary supplement. It is a medical food, a regulatory category that sits between those two, designed specifically for the nutritional management of kidney disease.
This KetoCitra review pulls together clinical trial data, ingredient analysis, real community feedback from Reddit and Facebook, and a full pricing breakdown so you can decide whether this product makes sense for your situation.
You won’t find marketing language here. You’ll find data, user experiences, and an honest assessment of where the evidence stands as of early 2026.
Santa Barbara Nutrients created KetoCitra based on research by Dr. Thomas Weimbs, a professor at the University of California, Santa Barbara (UCSB), who has studied polycystic kidney disease for over two decades [2].
The company operates as a Public Benefit Corporation, meaning it has a stated mission beyond profit. Dr. Weimbs serves as CEO of the company and continues to run a research lab at UCSB, a dual role worth keeping in mind as you evaluate the claims.
ADPKD affects more than 600,000 people in the United States alone [3]. It causes fluid-filled cysts to grow progressively in both kidneys, eventually impairing function and often leading to kidney failure.
Until recently, Tolvaptan (brand name Jynarque) was the only FDA-approved pharmacological option, and its side effects and cost make it a difficult choice for many patients.
KetoCitra offers a non-prescription alternative rooted in metabolic science. Whether that alternative delivers meaningful results is what this review examines.
KEY TAKEAWAYS
- KetoCitra delivers the ketone body BHB plus citrate in a sodium-free formula designed for CKD stages 1-3.
- A 2026 controlled clinical trial at Juntendo University in Tokyo found a 5.01% reduction in total kidney volume over 90 days in the KetoCitra + dietary education group, compared to an 8.03% increase in the control group [1].
- User testimonials report GFR improvements ranging from 10% to over 60% when combined with a ketogenic diet and intermittent fasting [4].
- Pricing ranges from $129/month (12-pack bulk) to $159 (one-time purchase), with a standard subscription at $139/month [5].
- Side effects are generally mild and limited to stomach upset, diarrhea, or bloating during the first few weeks [6].
- As a medical food, KetoCitra does not require FDA premarket approval, but all its ingredients carry GRAS (Generally Recognized as Safe) status [6].
- The strongest outcomes appear in users who pair KetoCitra with the Ren-Nu dietary program ($950 for 12 weeks) or an independent keto + intermittent fasting protocol [7].
- Several nephrologists, including physicians at the Cleveland Clinic, have recommended KetoCitra to patients based on the peer-reviewed research [8].
If this sounds like it could fit your situation, you can review the full product details at SantaBarbaraNutrients.com.
What Is KetoCitra and How Does It Work?
The Science Behind the Formula
Kidney cysts in ADPKD exhibit a metabolic defect that researchers have compared to the Warburg effect in cancer cells [9].
These cystic cells become dependent on glucose as their primary energy source and lose the ability to efficiently use fatty acids or ketone bodies. This glucose dependence fuels cyst growth and disease progression.
BHB = beta-hydroxybutyrate, a ketone body your liver produces during fasting or carbohydrate restriction. It serves as an alternative energy source for your cells. Supplementing with BHB shifts the metabolic environment away from glucose, effectively reducing the fuel supply for cystic cells.
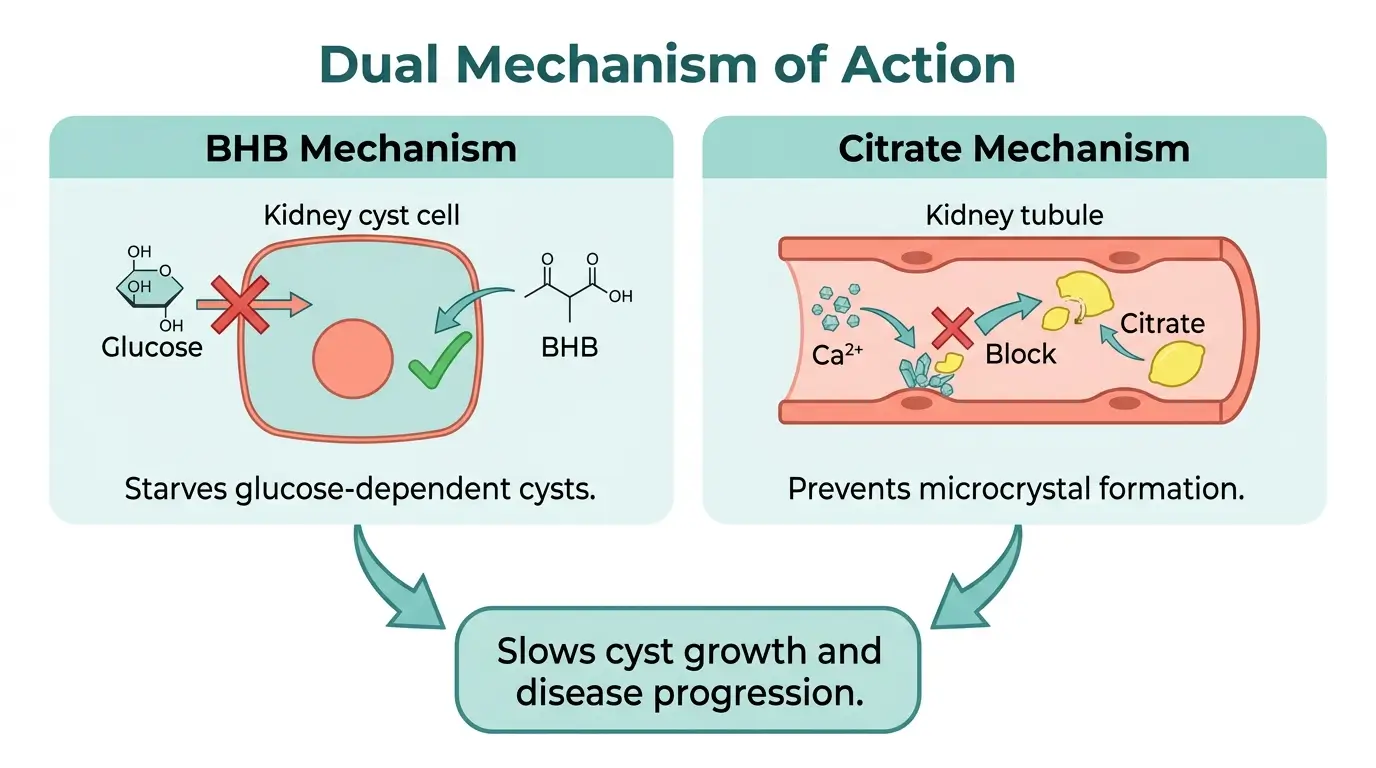
A 2019 study published in Cell Metabolism by Torres et al. demonstrated that BHB supplementation on a normal diet replicated the beneficial effects of a ketogenic diet on cyst growth in animal models of PKD [9].
This finding forms the scientific basis for KetoCitra’s BHB component.
The citrate component addresses a separate problem. People with ADPKD frequently have abnormally low levels of citrate in their urine, a condition called hypocitraturia [10].
Low urinary citrate increases the risk of calcium-based microcrystal formation in the kidneys. These microcrystals can trigger new cyst formation and accelerate disease progression [10].
Citrate binds with calcium in the urine to inhibit crystal formation, and it helps normalize urine pH, which is often too acidic in CKD patients.
Together, BHB and citrate address two distinct metabolic vulnerabilities in polycystic kidney disease simultaneously.
What’s Inside the Formula
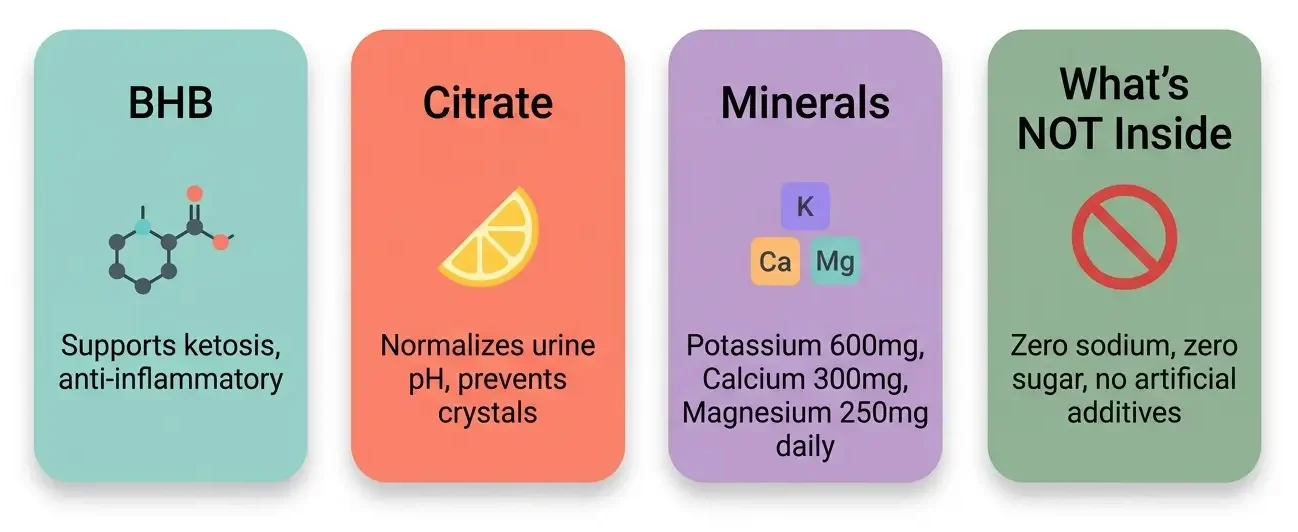
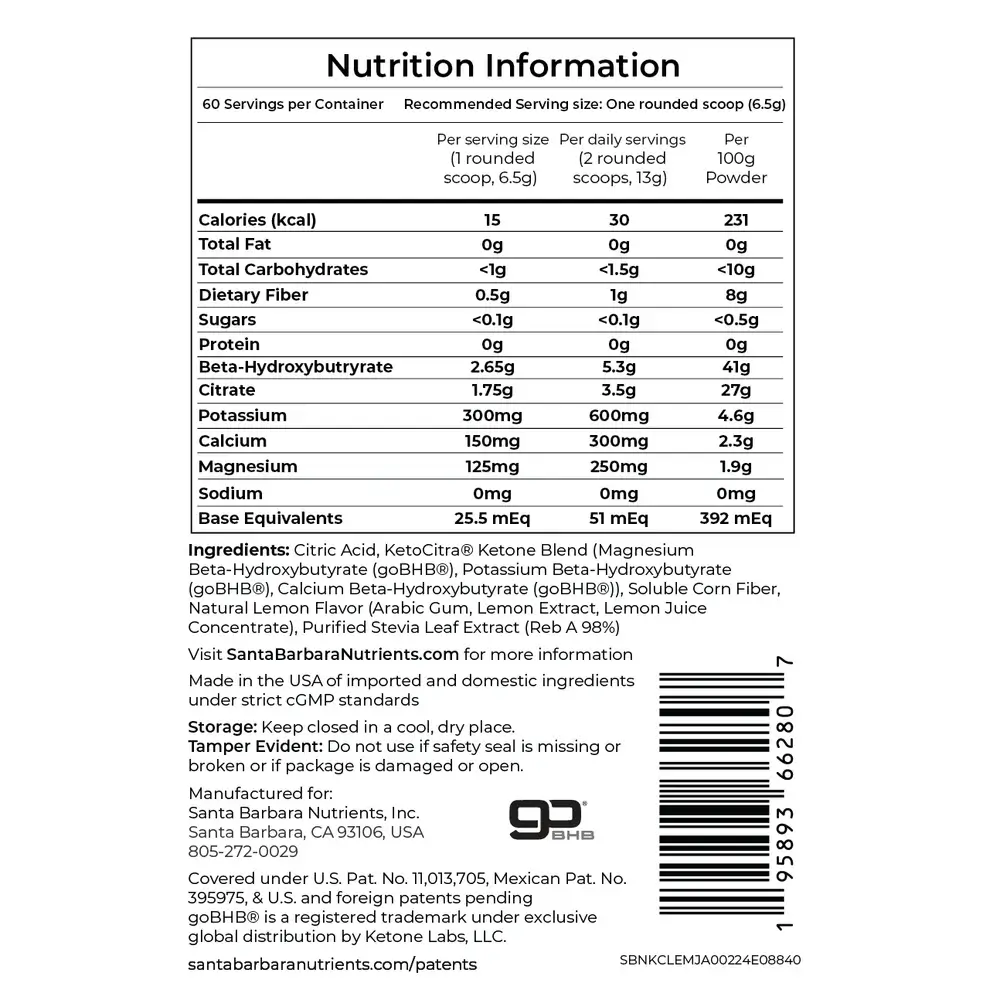
Each 6.5-gram serving of KetoCitra contains [11]:
- D,L-Beta-Hydroxybutyrate (BHB) delivered as potassium, calcium, and magnesium salts. The recommended dose is 2 servings per day, providing 600mg potassium, 300mg calcium, and 250mg magnesium daily.
- Citrate from citric acid, formulated to deliver approximately 50 mEq of alkali load per day at 2 servings [6].
The formula uses natural lemon flavoring (dehydrated lemon extract and lemon juice concentrate), stevia as a sweetener, and soluble corn fiber (non-GMO) as a flow agent. Each serving has 15 calories [11].
What’s NOT in the formula matters too.
KetoCitra contains zero sodium, zero sugar, no artificial flavors, no fillers, and no bulking agents. This is a deliberate design choice because dietary sodium intake accelerates disease progression in ADPKD [12].
Most over-the-counter BHB supplements use sodium-based salts, making them potentially harmful for kidney disease patients. KetoCitra replaces sodium with potassium, calcium, and magnesium.
Medical Food vs. Supplement vs. Drug
This distinction shapes how you should evaluate KetoCitra. The FDA regulates medical foods as a separate category defined in section 5(b) of the Orphan Drug Act [6].
Medical foods do not require premarket approval (similar to regular supplements). However, unlike supplements, medical foods must use only GRAS ingredients and must be intended for the dietary management of a specific disease under medical supervision.
Medical Food
Regulated by FDA for the dietary management of a specific disease. Used under medical supervision.
- All ingredients must be GRAS
- No FDA premarket approval required
- Can make disease-specific claims
- No prescription needed
Dietary Supplement
Intended for healthy individuals to promote normal body function. Not required to be GRAS.
- No GRAS requirement
- No FDA premarket approval
- Cannot make disease claims
- No prescription needed
Prescription Drug
Chemically synthesized compounds that must prove safety and efficacy before market approval.
- Typically NOT GRAS
- Rigorous FDA approval required
- Can make treatment claims
- Prescription required
What this means for you is that KetoCitra has a different evidence bar than a prescription drug. A prescription drug must prove safety and efficacy through rigorous clinical trials before it reaches the market.
A medical food does not. All KetoCitra’s ingredients are individually well-studied and recognized as safe, but the specific combination and its effects on PKD are still being validated through ongoing clinical trials.
The clinical evidence is growing, but it is not yet at the level of a fully approved drug. Keep this context in mind as you weigh the decision.
KetoCitra Pricing and Where to Buy
Cost is the most common concern in online discussions about KetoCitra. At $139 to $159 per month, it adds $1,668 to $1,908 to your annual healthcare spending. Bulk purchasing reduces the per-container cost.

| Purchase Option | Price / Container | Monthly Cost | Per Serving | Annual Cost |
|---|---|---|---|---|
| Option:One-Time Purchase | Price:$159 | Monthly:$159 | Per Serving:$2.65 | Annual:$1,908 |
| Option:Monthly Subscription Most Popular | Price:$139 | Monthly:$139 | Per Serving:$2.32 | Annual:$1,668 |
| Option:6-Pack Bulk | Price:$139 each | Monthly:$139 | Per Serving:$2.32 | Annual:$1,668 |
| Option:12-Pack Bulk | Price:$129 each | Monthly:$129 | Per Serving:$2.15 | Annual:$1,548 |
Each container provides a 30-day supply at the recommended 2 servings per day [5].
Santa Barbara Nutrients also offers a Starter Kit that bundles KetoCitra with a hydration bottle and pH test paper. The Ren-Nu dietary program, which is recommended alongside KetoCitra for best results, costs $950 for a 12-week program and includes 3 bottles of KetoCitra [13].
You can only purchase KetoCitra through SantaBarbaraNutrients.com and a small number of authorized retailers, including InKidney ($144) and Pounds Transformation ($135.99) [5].
It is not available on Amazon, Walmart, or any major pharmacy chain. International shipping costs a flat $20 [5].
(Ed. note: SBN openly suggests on their product page that you can subscribe at the lower price to try it once, then cancel without penalty. That level of transparency is uncommon.)
Is this cost justified? That depends on your financial situation and how you respond to the product. Some users in online forums have asked about DIY alternatives, and the honest answer is that you can buy separate BHB salts and potassium citrate for roughly $40-$80 per month.
However, generic BHB supplements typically contain sodium and are not formulated for kidney safety. The specific combination in KetoCitra, its sodium-free mineral profile, and the citrate alkaline load are what differentiate it from off-the-shelf options.
What Does the Clinical Research Say?
The 2026 Juntendo University Interim Trial
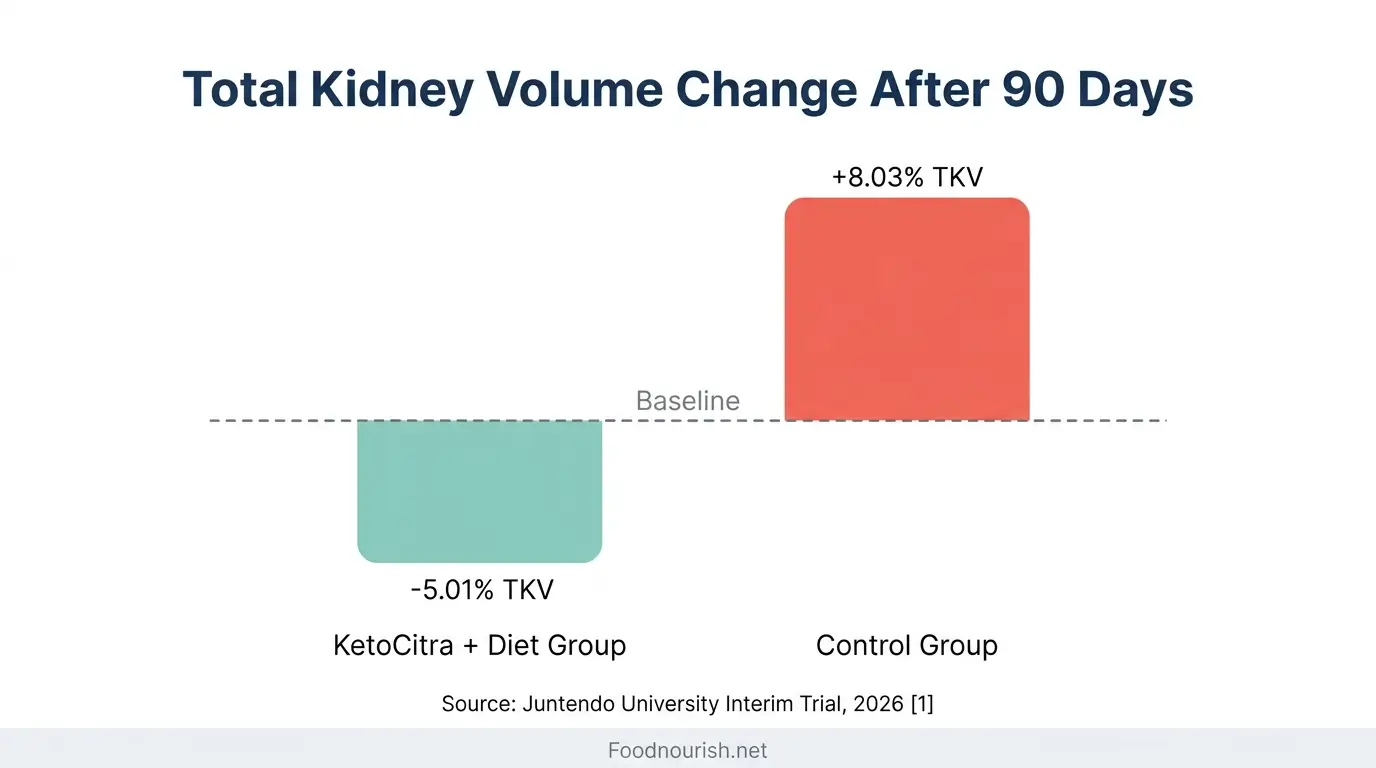
This is the most significant clinical data available for KetoCitra as of March 2026. Dr. Shigeo Horie at Juntendo University in Tokyo led a controlled intervention study testing KetoCitra combined with the Ren-Nu dietary education program in individuals with moderate ADPKD [1].
After only 90 days (this is a one-year trial with interim results), the intervention group experienced a statistically significant 5.01% reduction in total kidney volume (TKV). The control group, by contrast, showed an 8.03% increase in TKV over the same period [1].
That is a 13-point swing between the two groups in just three months.
The intervention group also showed a trend toward stabilization in eGFR (estimated glomerular filtration rate, a key measure of kidney function), with a +0.59 ml/min/1.73 m2 change, while the control group declined by -1.57 ml/min/1.73 m2 [1].
Perhaps the most notable finding was the 100% adherence rate with zero participants dropping out. This suggests the combined protocol of KetoCitra + dietary education is feasible and sustainable for patients [1].
These results were presented at the annual conference of the American Society of Nephrology and separately at the annual conference of the PKD Association of Japan [1].
The 2022 Ren-Nu Pilot Study
Bruen et al. published a retrospective case series in Kidney and Dialysis documenting outcomes from approximately two dozen individuals with ADPKD who completed the Ren-Nu program with KetoCitra [7].
This was NOT a controlled clinical trial. It was a beta test of the dietary program that reported qualitative outcomes, including reductions in kidney pain, headaches, and use of blood pressure medication.
While the self-reported results were encouraging, the study design limits the conclusions you can draw from it. There was no control group and no blinding.
The 2023 Animal Study
Torres et al. published a peer-reviewed study demonstrating that a combination of BHB and citrate led to partial disease regression in adult rats with established cystic disease [14].
The study found inhibition of cyst formation and reduced kidney injury markers. This paper has been cited 17 times in subsequent research, indicating the scientific community’s interest in the mechanism [14].
Animal results do not always translate directly to humans. However, this study provided the mechanistic foundation for the clinical trials now underway.
What the Research Does NOT Prove Yet
No large-scale, multi-center, long-term randomized controlled trial on KetoCitra has been completed. The Juntendo trial offers the strongest evidence so far, but it is still ongoing, and only 90-day interim data is available.
A 2024 review article in PMC specifically noted the gap, stating that “there are no clinical data supporting the use of supplements or nutrients such as KetoCitra” for PKD management [15].
This statement predates the 2026 interim trial results but reflects where the mainstream nephrology community stood.
“So does the clinical evidence justify the cost?” – The honest answer is that it depends on your risk tolerance and financial situation. The data is promising and growing, but it is early-stage. If you are waiting for rock-solid, drug-level clinical proof, that does not exist yet.
Where Does KetoCitra’s Evidence Stand?
KetoCitra currently sits at Level 3. A controlled clinical trial is underway with promising 90-day interim results, but the full 1-year data has not been published yet. The evidence is growing but not yet at the level of a fully validated pharmaceutical treatment.
What I Like About KetoCitra
- The research foundation is stronger than most dietary products on the market. Dr. Weimbs and his team at UCSB have published multiple peer-reviewed papers spanning animal models, pilot human studies, and now a controlled clinical trial. The trajectory is moving in the right direction, from bench to bedside, with data at each stage.
- The formulation itself reflects genuine thought about kidney safety. Zero sodium in a product designed for CKD patients is a meaningful design choice, especially when most competing BHB supplements use sodium as their primary mineral carrier. The inclusion of calcium and magnesium to bind dietary oxalate and phosphate adds a layer of kidney protection that goes beyond the BHB and citrate mechanism [6].
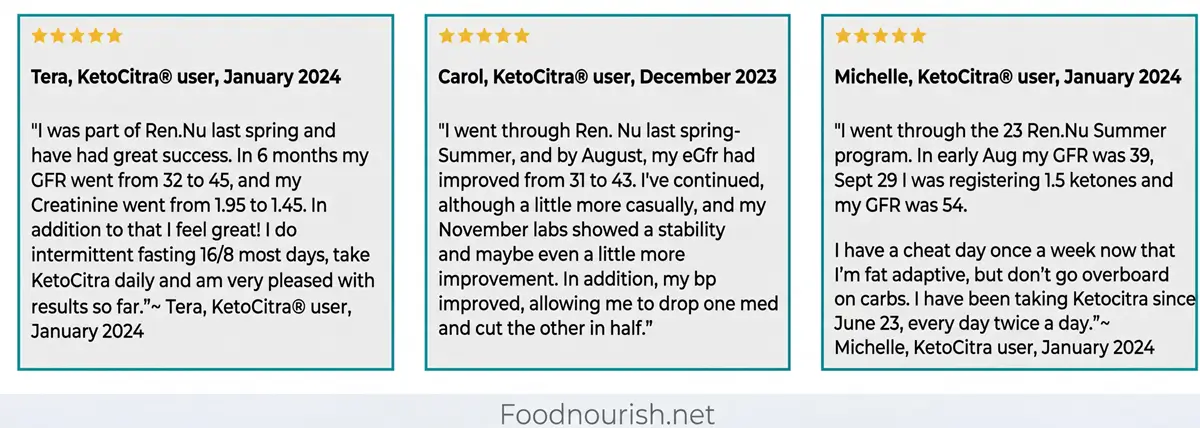
- User-reported outcomes are consistently positive for people who commit to the combined approach. Multiple testimonials describe GFR improvements of 10 to 30+ points over periods of 3 to 15 months, with some reporting dramatic results (GFR from 32 to 45 in six months, kidney volume reduced by approximately 30% in one year) [4].
- The taste gets almost universally positive reviews. Users describe it as a pleasant lemon flavor, easy to mix, and enjoyable to drink [4]. For a product that requires daily compliance over months or years, taste is not a trivial factor.
What I Don’t Like About KetoCitra
- The cost is a real barrier. At $1,548 to $1,908 per year for KetoCitra alone, and potentially $2,618 or more when you add the Ren-Nu program, this is a significant ongoing expense with no insurance coverage. One Facebook group member put it bluntly, posting “I need more ketocitra, but the cost is prohibitive” [16].
- The strongest results come from users who combine KetoCitra with a ketogenic diet and intermittent fasting. If you take KetoCitra alone without dietary changes, you are likely getting the citrate benefits (urine pH normalization, stone prevention) and some BHB, but you may not achieve the metabolic ketosis state that appears to drive the most meaningful kidney outcomes.
- The clinical trial that produced the 5% TKV reduction paired KetoCitra with a full dietary education program [1]. Separating the contribution of KetoCitra alone from the dietary changes is difficult based on current data.
- The dual role of Dr. Weimbs as both the lead researcher and CEO of Santa Barbara Nutrients creates a potential conflict of interest. This does not invalidate the research. The Juntendo trial, for instance, was led by an independent investigator in Japan. But it does mean that the primary source of information about KetoCitra is also the entity profiting from its sale.
- One Reddit commenter observed that “99% of the information when googling is from the Santa Barbara Nutrients website” [8]. That information asymmetry should make you more, not less, careful in evaluating the claims.
- KetoCitra is not appropriate for advanced kidney disease (CKD stages 4-5) because of its potassium content [6]. Individuals with impaired renal function may not be able to excrete potassium efficiently, leading to dangerous hyperkalemia.
If you have advanced CKD, you need your physician to evaluate whether KetoCitra is safe for you.
What Real Users Say on Reddit, Facebook, and YouTube
Positive Experiences
The most common positive report across platforms involves GFR stabilization or improvement.
One testimonial described a jump from GFR 54 to GFR 60 after returning to KetoCitra following a 2-3 month period using a different product [4].
Another user documented a move from GFR 27 to 44 in “a couple of months” using clean keto, intermittent fasting, and KetoCitra twice a day [4].
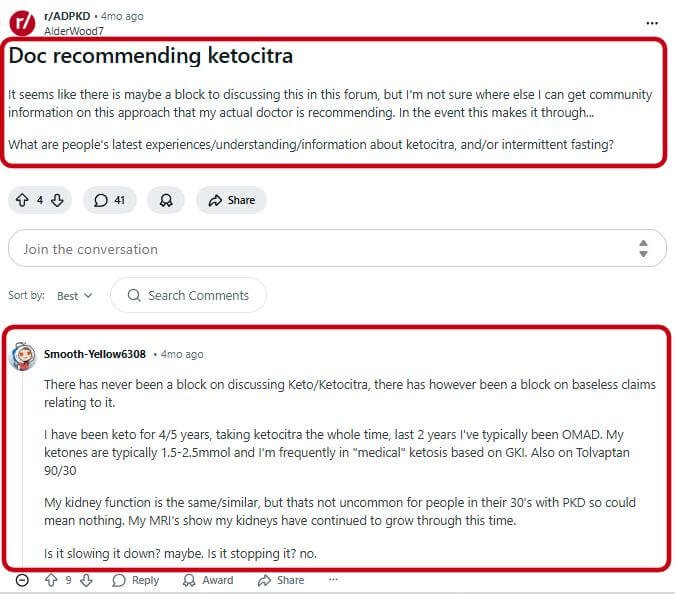
On the Reddit r/ADPKD forum, one commenter shared that their physician at the Cleveland Clinic “peer reviewed the research and said it was some of the best research he has ever seen in PKD” [8].
Another Reddit user who identified as someone on Tolvaptan for 17+ years reported that adding KetoCitra helped keep creatinine “remarkably stable” at 1.2 at age 55 [4].
Blood pressure reduction appears frequently in testimonials. One user reported going from struggling to stay below 140/85 down to 120/80 after starting KetoCitra, and was able to reduce medication dosage [4].
Another reported dropping one blood pressure medication entirely and cutting the other in half [4].
Reduced kidney pain is another recurring theme. Multiple users across Facebook and YouTube specifically mention that flank pain decreased significantly after starting KetoCitra, particularly when combined with dietary changes [4].
Mixed and Critical Feedback
Not everyone reports positive results. On Reddit, one user stated plainly, “I’ve been taking ketocitra for a year with keto diet. My kidney function is still declining” [8].
MRI data from another long-term user showed that while overall health improved, kidneys continued to grow, raising the question of whether KetoCitra merely slows progression or actually halts it [8].
A Reddit commenter who identified as a Wall Street executive offered a candid cost-benefit analysis, calling KetoCitra “effective as a citrate” for preventing kidney stones but “pretty ineffective at raising ketone levels.”
The user concluded that if you’re not price-sensitive, “it won’t hurt, and may help a little,” but if money is tight, “probably not worth it” [8].
Another commenter pointed out that you can supplement the individual ingredients (magnesium citrate, calcium citrate, potassium citrate) separately at a lower cost, calling it “very easy to do” [8]. This is a valid point, though generic BHB supplements typically contain sodium, which KetoCitra deliberately avoids.
The Pattern That Emerges
Users who combined KetoCitra with a ketogenic diet AND intermittent fasting reported the strongest results. Users who took KetoCitra alone without dietary changes reported more modest outcomes. And across all communities, a consistent theme appeared when users shared lab results with their nephrologists, the doctors who were initially skeptical became increasingly supportive.
“Will my nephrologist approve of this?” Based on community feedback, expect initial caution followed by openness if your lab numbers improve.
Want to see if your experience matches these reports? You can learn more about KetoCitra at SantaBarbaraNutrients.com.
KetoCitra Side Effects and Safety
Side Effects to Know
- Stomach upset
- Diarrhea
- Constipation
- Stomach pain
Most common in first-time users or when starting at a high serving size. Start with a lower dose and increase gradually.
Santa Barbara Nutrients reports that some users experience stomach upset, diarrhea, constipation, or stomach pain [6].
These side effects occur most often in first-time users or when someone starts at a high serving size. The company recommends starting with a lower dose and increasing gradually.
The potassium content (600mg daily at 2 servings) creates a specific safety concern for anyone with hyperkalemia or severely impaired renal function [6].
Your physician should check your blood electrolyte levels before you start using KetoCitra, especially if you have CKD stage 3 or beyond.
KetoCitra is manufactured under cGMP (current Good Manufacturing Practice) standards in an NSF-certified, FDA-registered facility [6].
Quality control involves both in-house and independent third-party laboratory testing. All ingredients carry GRAS status as required by FDA regulations for medical foods.
One safety advantage over generic BHB products is the absence of sodium. If you have CKD and are trying to limit sodium intake to below 2,300mg per day (as most nephrologists recommend), adding a sodium-based BHB supplement would work against that goal. KetoCitra sidesteps this problem entirely [11].
KetoCitra vs. Tolvaptan and Other Approaches
If you have ADPKD, you are likely evaluating several options simultaneously. This comparison captures the key differences.
| Category | KetoCitra | Tolvaptan (Jynarque) | DIY BHB + Citrate | Keto Diet Alone |
|---|---|---|---|---|
| Type | Medical food | Prescription drug | OTC supplements | Dietary intervention |
| Monthly Cost | $129-$159 | $1,000-$3,000+ (varies by insurance) | $40-$80 | $0 (food cost shifts) |
| Prescription | No | Yes | No | No |
| Mechanism | Exogenous BHB + citrate supplementation | Blocks vasopressin (reduces cyst fluid) | Exogenous ketones + citrate (often sodium-based) | Endogenous ketosis via carb restriction |
| Clinical Evidence | 1 interim controlled trial + animal studies | Multiple large RCTs | No kidney-specific trials | 1 RCT (KETO-ADPKD, 2023) |
| Side Effects | Mild GI issues (temporary) | Frequent urination, liver risk (<3%), thirst | Sodium load harmful to kidneys | Adherence difficulty, potential deficiencies |
| Best For | CKD 1-3, non-pharmaceutical support | Moderate-to-severe PKD | Budget-conscious patients | Highly motivated individuals |
Some users combine multiple approaches. Several Reddit commenters reported taking both Tolvaptan and KetoCitra simultaneously, along with intermittent fasting [8].
No contraindication between these approaches has been reported, but you should discuss any combination therapy with your nephrologist.
The Tolvaptan comparison deserves extra context. One Reddit thread raised questions about Tolvaptan’s efficacy data, with a user noting that some scientists have questioned whether Tolvaptan’s GFR benefits could be partially explained by muscle mass loss rather than true kidney function preservation [8].
A separate theory suggests that Tolvaptan’s benefits may stem partly from the massive water intake it forces, not from the drug mechanism itself. A water intake study is reportedly underway to test this hypothesis [8].
These are unresolved questions in the PKD community, but they illustrate why patients are exploring metabolic alternatives like KetoCitra.
Can You Take KetoCitra Without the Keto Diet?
Yes. Santa Barbara Nutrients states on their website that “KetoCitra can be taken by itself without any other changes to the normal diet” [6].
However, the results will likely differ. When you take KetoCitra without carbohydrate restriction, you get the citrate benefits (normalized urine pH, reduced microcrystal risk, stone prevention) and a modest boost in circulating BHB. You do NOT get the sustained nutritional ketosis that appears to drive the most significant kidney outcomes in the research.
The 2026 Juntendo trial paired KetoCitra with a full ketogenic dietary education program [1].
Separating the specific contribution of KetoCitra from the dietary changes is impossible based on the current study design.
The animal studies used BHB supplementation on a normal diet and still showed benefits [9], which suggests KetoCitra provides some value independently. But the human data showing the strongest results always involved dietary modifications.
“What if I can’t do a full keto diet?”
A reasonable middle ground is pairing KetoCitra with intermittent fasting (16:8 or similar). Several users reported positive outcomes with this less restrictive combination [4].
The Ren-Nu program itself is designed as a phased approach over 12 weeks, gradually introducing ketogenic eating rather than requiring immediate strict adherence [13].
Who Should and Shouldn’t Use KetoCitra
May Be Right For You
- CKD stages 1-3, especially ADPKD
- Already on or considering a ketogenic diet
- Seeking non-pharmacological kidney support
- History of kidney stones (need citrate)
Probably Not Right For You
- CKD stages 4-5 (unless physician approved)
- High blood potassium or impaired electrolytes
- Looking for a standalone cure for PKD
- Cannot sustain $129-$159/month long-term
KetoCitra may be appropriate if you have CKD stages 1-3 (especially ADPKD), if you are already on or considering a ketogenic diet, if you want non-pharmacological kidney support, or if you have a history of kidney stones and need citrate supplementation.
KetoCitra is NOT appropriate if you have CKD stages 4-5 (unless your physician specifically approves it), if you have hyperkalemia or impaired electrolyte regulation, if you are looking for a standalone cure for PKD (it is a dietary management tool), or if the ongoing monthly cost would create financial strain without a realistic path to evaluate whether it’s working for you within 3-6 months.
If you fall into the appropriate category, work with your nephrologist to establish baseline bloodwork before starting. Track your eGFR, creatinine, and blood electrolytes over time. Give the product at least 3 months before evaluating results, based on the timeline reflected in both the clinical trial and user testimonials.
Frequently Asked Questions
How We Researched This KetoCitra Review
This review analyzed over 25 sources spanning peer-reviewed research, manufacturer documentation, and community feedback.
Sources included the Santa Barbara Nutrients website (product pages, FAQ, ingredient profile, testimonials, blog posts, and the February 2026 clinical trial press release), three peer-reviewed publications (Torres et al. 2023, Bruen et al. 2022, and the 2025 ASN conference abstract from the Juntendo trial), the UC Santa Barbara MCDB news release from 2021, Reddit r/ADPKD discussions from 2021 through 2025, Facebook Weimbs Lab community posts from 2022 through 2024, YouTube testimonial videos, and current pricing data from SantaBarbaraNutrients.com, InKidney.com, and PoundsTransformation.com.
Selection criteria focused on English-language sources with verifiable authorship or platform authentication. The data covers November 2021 (KetoCitra’s launch) through March 2026.
Key limitations of this review include the following.
- No personal hands-on product testing was conducted.
- User testimonial data is self-reported and not independently verified.
- The Juntendo clinical trial is ongoing, and only 90-day interim data is available.
- Santa Barbara Nutrients is the primary source for most KetoCitra information, creating an inherent information asymmetry that an independent reviewer cannot fully resolve.
- The community feedback aggregated from Reddit and Facebook represents a self-selected population, meaning people with strong positive or negative experiences are more likely to post than those with neutral outcomes.
Citations
- [1] Santa Barbara Nutrients, “Interim Clinical Trial Results Show Significant Efficacy of KetoCitra”, February 18, 2026. Conference abstract available via JASN/ASN 2025.
- [2] UC Santa Barbara MCDB, “KetoCitra is Here”, November 29, 2021.
- [3] Santa Barbara Nutrients, “KetoCitra FAQs”, referencing ADPKD prevalence data.
- [4] Santa Barbara Nutrients, “KetoCitra Testimonials: Real Stories of Kidney Health”, accessed March 2026.
- [5] Santa Barbara Nutrients, KetoCitra Product Page, accessed March 2026. Pricing also verified via InKidney and Pounds Transformation.
- [6] Santa Barbara Nutrients, “KetoCitra FAQs: Kidney Health Nutrition”, accessed March 2026.
- [7] Bruen DM, Kingaard JJ, Munits M, et al., “Ren.Nu, a Dietary Program for Individuals with Autosomal-Dominant Polycystic Kidney Disease”, Kidney and Dialysis, 2022;2:183-203.
- [8] Reddit r/ADPKD, “Doc recommending ketocitra”, November 21, 2025, 41 comments.
- [9] Torres JA, Kruber S, Barber E, et al., “Ketosis Ameliorates Renal Cyst Growth in Polycystic Kidney Disease”, Cell Metabolism, 2019.
- [10] Santa Barbara Nutrients, “What’s in KetoCitra?”, November 22, 2023.
- [11] Santa Barbara Nutrients, “Ingredient & Nutrient Profile of KetoCitra”, accessed March 2026.
- [12] Chebib FT, Torres VE, “Recent Advances in the Management of Autosomal Dominant Polycystic Kidney Disease,” Clin J Am Soc Nephrol, 2018. PMID: 30049849.
- [13] Ren-Nu Program, Ren-Nu.org, accessed March 2026. Pricing from Instagram announcement, August 2025.
- [14] Torres JA, et al., “A combination of beta-hydroxybutyrate and citrate ameliorates disease progression in a rat model of polycystic kidney disease”, 2023. PMC11207547. Cited by 17 subsequent publications.
- [15] PMC, “Polycystic Kidney Disease Diet: What is Known and What is Safe”, 2024.
- [16] Facebook, Weimbs Lab Community Group, “I need more ketocitra, but the cost is prohibitive”, October 17, 2024.
- [17] Science Daily, “Clinical trial demonstrates that the ketogenic diet is effective at controlling polycystic kidney disease”, December 11, 2023.
You can share and adapt this content for non-commercial purposes, provided you give appropriate credit.
